
Principal Investigator
Technology
Professor
Department of Cell Biology, Graduate School of Medicine, The University of Tokyo
Technology Development for In Vivo Imaging of Cell Dynamics
Research
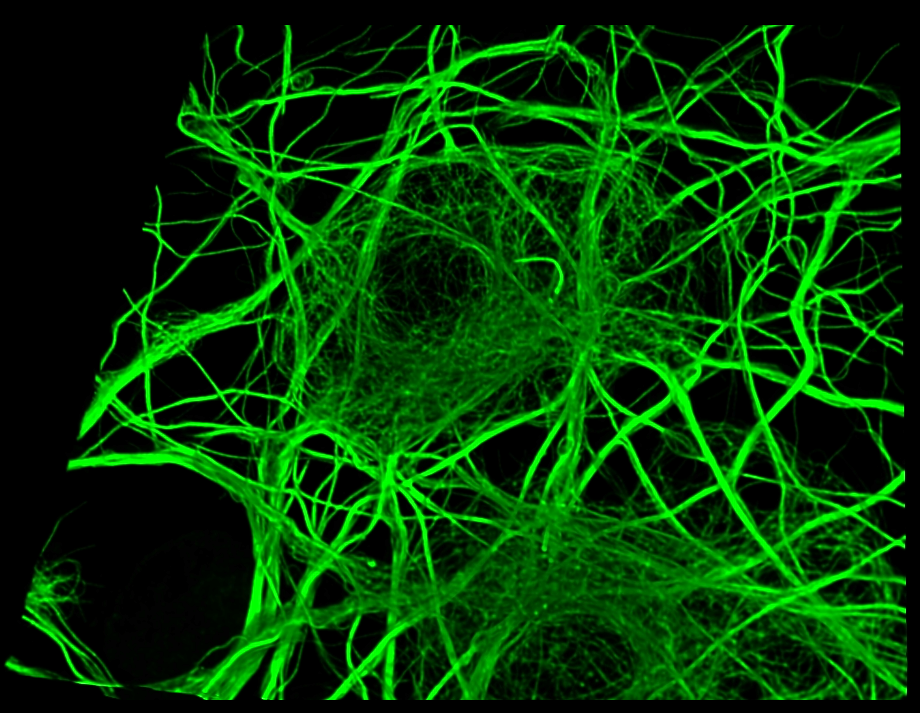
Our laboratory aims to deepen our understanding of the fundamental question, "What is life?" While recent advancements in molecular and structural biology have significantly advanced our understanding of life phenomena, this essential question remains unanswered.
The hallmark of our approach is the direct observation of life phenomena at the molecular level within living cells, rather than relying on imaginary illustrations or static models. To realize this vision, we are focusing on three key areas of technological development:
1. High-resolution live-cell imaging techniques: Development of cutting-edge microscopy technologies to directly observe molecules functioning within living cells.
2. Cell state visualization probes: Design and synthesis of innovative molecular probes to visualize dynamic changes in cellular states.
3. Image analysis methods: Development of mathematical and computational methods to extract valuable information from microscopic images.
By integrating these technologies and applying them to the observation of neurons, we aim to contribute to the elucidation of brain function and mechanisms of neurological disorders.
Our research is positioned at the intersection of biology, physics, chemistry, and information science. Through this interdisciplinary approach, we strive to open new frontiers in life sciences.
Publications
Kondratiev AY, Inutsuka Y, Okada Y. Continuous cell cycle representation using ordinal regression and siamese network from quantitative phase images. ISBI2024, 2024.
Wu Q, Taki M*, Tanaka Y, Kesherwani M, Phung QM, Enoki S, Okada Y*, Tama F*, Yamaguchi S*. Stereochemistry-Dependent Labeling of Organelles with a Near-Infrared-Emissive Phosphorus-Bridged Rhodamine Dye in Live-Cell Imaging. Angewandte Chemie International Edition, 63: e202400711, 2024.
Ando R, Shimozono S, Ago H, Takagi M, Sugiyama M, Kurokawa H, Hirano M, Niino Y, Ueno G, Ishidate F, Fujiwara T, Okada Y, Yamamoto M, Miyawaki A. StayGold variants for molecular fusion and membrane-targeting applications. Nat Methods. 2023.
Ishikawa S, Iwanaga Y, Ueyama T, Li X, Hojo H, Fujinaga I, Katashima T, Saito T, Okada Y, Chung U, Sakumichi N, Sakai T. Percolation-induced gel–gel phase separation in a dilute polymer network. Nat. Mater. 22: 1564–1570, 2023.
Okamoto K, Fujita H, Okada Y, Shinkai S, Onami S, Abe K, Fujimoto K, Sasaki K, Shioi G, Watanabe TM. Single-molecule tracking of Nanog and Oct4 in living mouse embryonic stem cells uncovers a feedback mechanism of pluripotency maintenance. EMBO J. 42: e112305, 2023.
Fukuda T, Furukawa K, Maruyama T, Yamashita SI, Noshiro D, Song C, Ogasawara Y, Okuyama K, Alam JM, Hayatsu M, Saigusa T, Inoue K, Ikeda K, Takai A, Chen L, Lahiri V, Okada Y, Shibata S, Murata K, Klionsky DJ, Noda NN, Kanki T. The mitochondrial intermembrane space protein mitofissin drives mitochondrial fission required for mitophagy. Mol Cell. 83: 2045-2058.e9, 2023.
Nozaki T, Shinkai S, Ide S, Higashi K, Tamura S, Shimazoe MA, Nakagawa M, Suzuki Y, Okada Y, Sasai M, Onami S, Kurokawa K, Iida S, Maeshima K. Condensed but liquid-like domain organization of active chromatin regions in living human cells. Science Advances. 9: eadf1488, 2023.
Katoh TA, Omori T, Mizuno K, Sai X, Minegishi K, Ikawa Y, Nishimura H, Itabashi T, Kajikawa E, Hiver S, Iwane AH, Ishikawa T, Okada Y, Nishizaka T, Hamada H. Immotile cilia mechanically sense the direction of fluid flow for left-right determination. Science. 379: 66-71, 2023.
Nagao Y, Sakamoto M, Chinen T, Okada Y, Takao D. Robust classification of cell cycle phase and biological feature extraction by image-based deep-learning. Mol Biol Cell 31: 1346-1354, 2020.
Fujioka Y, Alam JM, Noshiro D, Mouri K, Ando T, Okada Y, May AI, Knorr RL, Suzuki K, Ohsumi Y, Noda NN. Phase separation organizes the site of autophagosome formation. Nature 578: 301-305, 2020.
Biography
Graduated from the University of Tokyo, Faculty of Medicine in 1993. Medical Doctor (M.D.) and Ph.D. After serving as a JSPS Research Fellow and Assistant Professor at the same university's graduate school, became a Team Leader at RIKEN Center for Biosystems Dynamics Research (formerly known as RIKEN Quantitative Biology Center until reorganization in 2018) in 2011. Since 2016, Professor at the Department of Physics, Graduate School of Science, The University of Tokyo (concurrent position with RIKEN). Since 2020, Professor at the Department of Molecular and Cellular Biology, Graduate School of Medicine, The University of Tokyo (concurrent positions with RIKEN and Graduate School of Science).
Specializes in cell biology and biophysics. Conducts research utilizing various imaging technologies, with a focus on optical microscopy (single-molecule imaging, super-resolution microscopy techniques).


